Almost five years ago, the Food and Drug Administration (FDA) approved the only permanent heart implant proven to reduce stroke risk in people with atrial fibrillation not caused by a heart valve problem, or non-valvular Afib. Today, this lifesaving alternative, the Watchman Device, has changed the lives of over 75,000 patients. Now, the next-generation of Watchman, the Watchman FLX, has just received the CE mark in Europe.
A Lifesaving Alternative: The Watchman Device
The Watchman, is a left atrial appendage closure (LAAC) implant about the size of a quarter, designed to reduce lifelong risk of stroke in patients with a history of non-valvular Afib, as a stroke prevention alternative to long-term blood thinners. The device is placed in the patient’s left atrial appendage (LAA) to permanently close off this part of the heart where 90 percent of stroke-causing clots are formed.
People with a history of non-valvular Afib are five times more likely to suffer a stroke than someone with a regular heartbeat. While previously treated with blood thinners only, patients now have the option of a one-time implantation procedure that effectively reduces the risk of stroke.
Watchman Implantation Procedure
The Watchman Implant procedure takes no longer than 1-2 hours and requires just one overnight hospital stay. During the procedure, the patient undergoes general anesthesia and a narrow tube is inserted via a small incision in the upper leg. The doctor then guides the Watchman by the narrow tube up into the LAA of the heart. After the procedure, the patient will continue taking a blood thinner for 45 days or until heart tissue has grown over the implant, permanently closing off the LAA.
The minimally invasive procedure comes with a safe and speedy recovery, no known side effects, and lifelong peace of mind knowing that you are no longer at risk for potential bleeding caused by long-term blood thinners, and more importantly, no longer at risk for a stroke.
The Next Generation of Watchman
The first version of Watchman became internationally available in 2009. Now a new generation of the technology, called Watchman FLX, has been designed for simplified implantation to fit a wider range of patients. The new Watchman FLX device allows for more flexibility during the implantation procedure, with a fully enclosed, rounded frame to enhance the closing of the LAA. The new design will allow doctors to fully recapture and reposition the device during the procedure, making it more customizable to the individual patient’s anatomy.
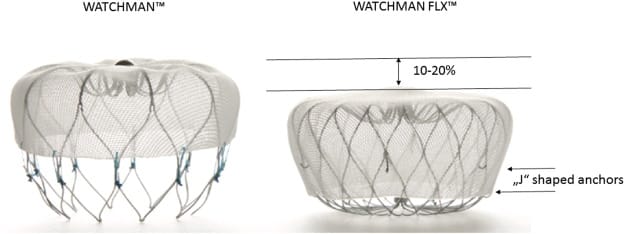
The Watchman FLX is not available for sale in the United States yet. However, Boston Scientific has announced that the new device has received CE mark and they have begun a limited market release in Europe. The company plans to expand marketization to additional sites for the remainder of 2019.
“The robust clinical evidence and successful commercial outcomes of the Watchman device to-date reinforce the value of this procedure for all appropriate patients,” said Kevin Ballinger, President of Interventional Cardiology.
Is the Watchman Right for You?
If you have non-valvular Afib and have been recommended for a blood thinner, the Watchman may be the right choice for you. To learn more about the Watchman device and to see if the procedure is right for you, please contact CVG online or visit one of our locations today.













